論文がアクセプトされ裏表紙を飾りました(綱島先生)

Chemo-Chromism in an Orthogonal Dabco-Based Co(II) Network Assembled by
Methanol-Coordination and Hydrogen Bond Formation
M. Shiga, S. Kawaguchi, M. Fujibayashi, S. Nishihara, K. Inoue, T. Akutagawa,
S. Noro, T. Nakamura, R. Tsunashima
Dalton Trans., 2018, 47, 7656-7662.
DOI: 10.1039/c8dt01220b
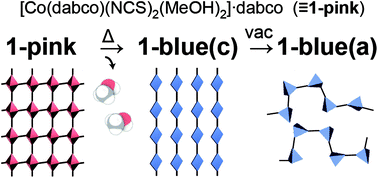
Abstruct: Newly prepared single crystals of [Co(II)(dabco)(NCS)2(MeOH)2]·dabco
(1-pink; dabco: 1,4-diazabicyclo[2.2.2]octane) showed chromotropic behaviour
in the solid state, changing from pink to blue upon heating or grinding.
The complex 1-pink exhibited a two-dimensional orthogonal network structure
with the coordination chain of –dabco–Co– bridged by hydrogen bonds between
coordinative methanol and a second dabco molecule, where the methanol molecule
was trapped by coordinative and hydrogen bonds. Chromism was demonstrated
to stem from the quantitative desorption of methanol from 1-pink to produce
[Co(II)(dabco)(NCS)2]·dabco (1-blue(c)) by thermogravimetric (TG) and temperature
controlled gas chromatography-mass spectrometry (GC-MS) analyses, and powder
X-ray diffraction (XRD) analysis suggests that the transformation between
the crystalline phases of 1-pink and 1-blue(c) occurred with similar lattice
parameters. Furthermore, the desolvated species showed chemo-chromic behaviour
due to the selective size- and polarity-dependent adsorption of solvent
molecules.