論文がアクセプトされました(村藤先生・上條先生・石黒先生 共著)
Blue Amino Acids Derived from Azulen-1-ylboronic Acid Pinacol Ester via
the Petasis Reaction
T. Murafuji, Y. Tasaki, M. Fujinaga, K. Tao, S. Kamijo, K. Ishiguro
Synthesis, 2017, 49, 1037-1042.
DOI: 10.1055/s-0036-1588907
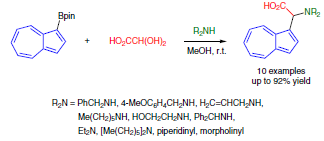
Abstruct: Azulen-1-ylboronic acid pinacol ester undergoes a three-component Petasis
reaction with amines and glyoxylic acid hydrate to give azulenylglycine
derivatives in good yields. The progress of the reaction is indicated by
a characteristic color change from violet to blue due to the altered π-conjugation
of the azulene chromophore. The azulenylboronic ester is more reactive
than its phenyl counterpart and even 2-styryl- and 2-thienylboronic pinacol
esters, which have a strong electron-donating organyl group on boron. These
results reflect the unique π-electron system of non-alternant azulene.